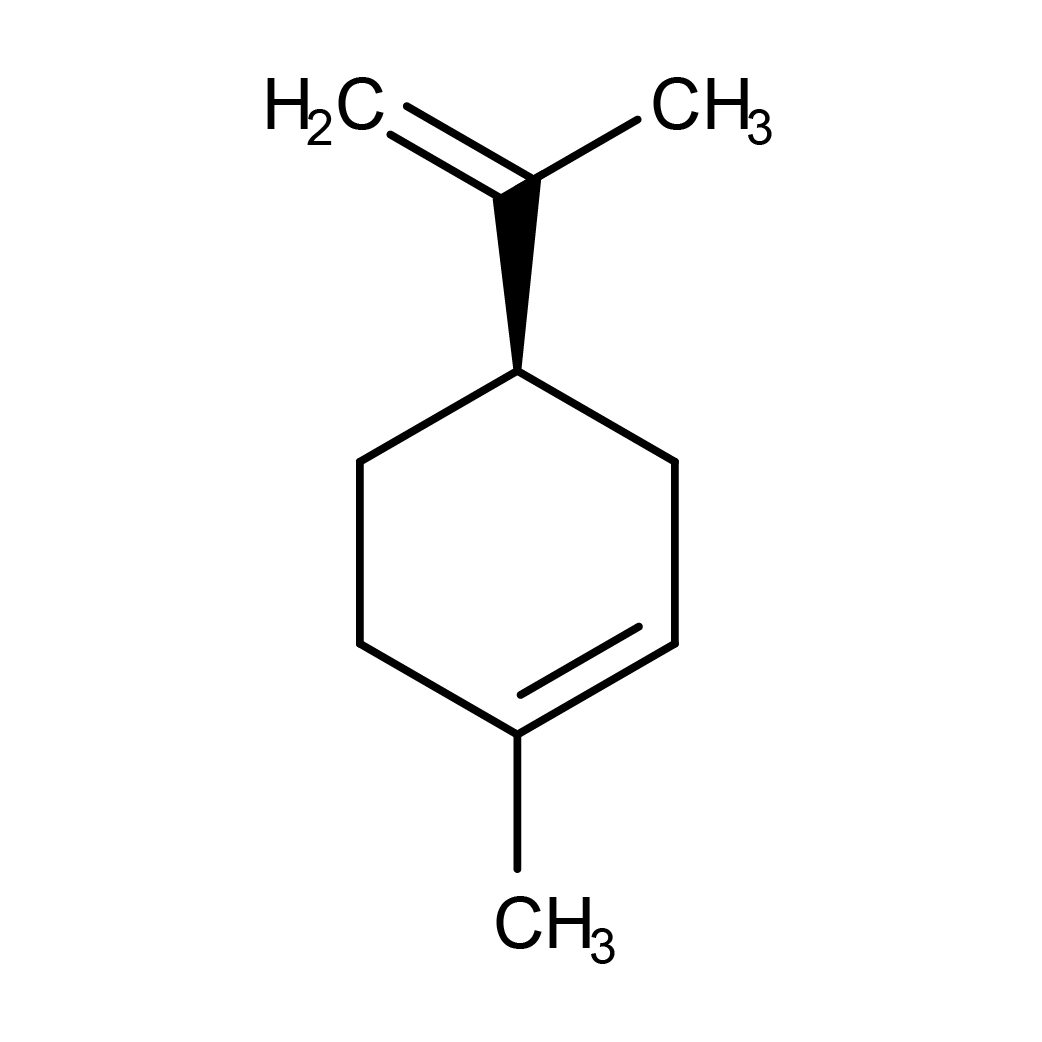
D-Limonene
Synonyms: D-Limonene, "(+)-Limonene", "(R)-(+)-Limonene", "(+)-carvene", "(+)-(4R)-Limonene", "Citrene", "(4R)-Limonene", "(+)-p-Mentha-1,8-diene", "D-(+)-Limonene", "(R)-4-Isopropenyl-1-methyl-1-cyclohexene", "(R)-Limonene", "Glidsafe", "Refchole", "Carvene", "Kautschiin", "Glidesafe", "(+)-R-Limonene", "(R)-p-Mentha-1,8-diene", "D-Limonen", "d-p-Mentha-1,8-diene", "Limonene, D-", "(D)-Limonene", "(+)-Dipentene", "Biogenic SE 374", "(+)-alpha-Limonene"
Source: D-limonene is one of the most common terpenes in nature. It is a major constituent in several citrus oils (orange, lemon, mandarin, lime, and grapefruit).
Identifiers:
IUPAC Name: (4R)-1-methyl-4-prop-1-en-2-ylcyclohexene
CAS Number: 5989-27-5
PubChem ID: 440917
InChiKey: XMGQYMWWDOXHJM-JTQLQIEISA-N
Canonical SMILES: CC1=CCC(CC1)C(=C)C
Structural Properties:
Molecular Formula: C10H16
Molecular Weight: 136,230
Pharmacophore Features:
Number of bond donors: 0
Number of bond acceptors: 0
Number of atoms different from hydrogen: 10
Downloads
2D structure (.sdf)
3D structure (.sdf)
3D structure (.mol2)
3D structure (.pdb)
3D structure (.pdbqt)
Search Similar molecules
Evidence Supporting This Chemical as an Emerging Contaminant

Baldwin, A. K., Corsi, S. R., De Cicco, L. A., Lenaker, P. L., Lutz, M. A., Sullivan, D. J., & Richards, K. D. (2016). Organic contaminants in Great Lakes tributaries: Prevalence and potential aquatic toxicity. Science of the Total Environment, 554, 42-52.
External Links

2D-structure
3D-structure